 
The International Union of Pure and Applied Chemistry designated the isotope C-12 as the basis for atomic weights, while the unstable isotope C-14, with a half-life of 5700 years, is used for carbon dating. How to Write the Electron Configuration for Carbon Carbon is the sixth element with a total of 6 electrons. Carbon-based compounds are the basis for all living systems and comprise the structure of fossil fuels in the form of hydrocarbons. There are two electrons in the first shell (both in the 1s orbital), and 4 electrons in the second shell (the 2s and 2p orbitals). There are 4 valence electrons but only 2 electrons are unpaired in. The first electron has the same four quantum numbers as the hydrogen atom electron ( n 1, l 0, ml 0, m s + 1 2 ). If we consider carbon in the ground state, its electronic configuration will be 1s2 2s2 2p2. The helium atom contains two protons and two electrons. Carbon’s small size allows it to form multiple bonds with many other small atoms, including carbon atoms, and is prevalent in a large number of chemical compounds. The electron configuration and the orbital diagram are: Following hydrogen is the noble gas helium, which has an atomic number of 2. Carbon Electron Configuration is an element of the periodic table and its atomic number is 6. According to the Aufbau principle, electrons will fill the lowest energy level sub. 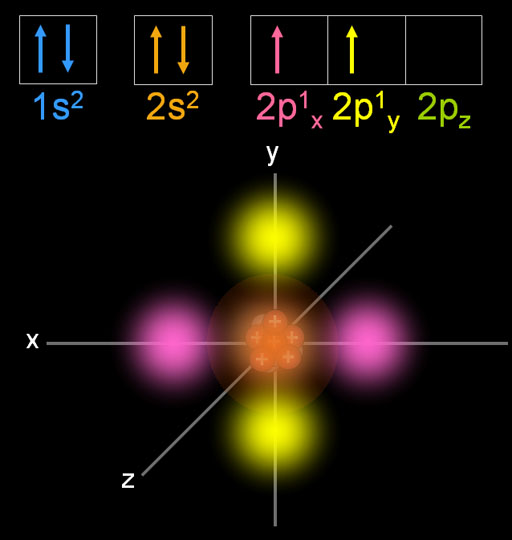
Four allotropes of carbon are known: amorphous (e.g., charcoal and soot), graphite, diamond, and fullerenes. Carbon has a proton number of 6, meaning that it also contains six electrons. Obtained from: burning with insufficient oxygenĬarbon has been known since ancient times when it was produced by burning organic material in the presence of insufficient oxygen. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
